The factor-label method yields the desired cancellation of units, and the computed result is on the order of 10 22 as expected. 
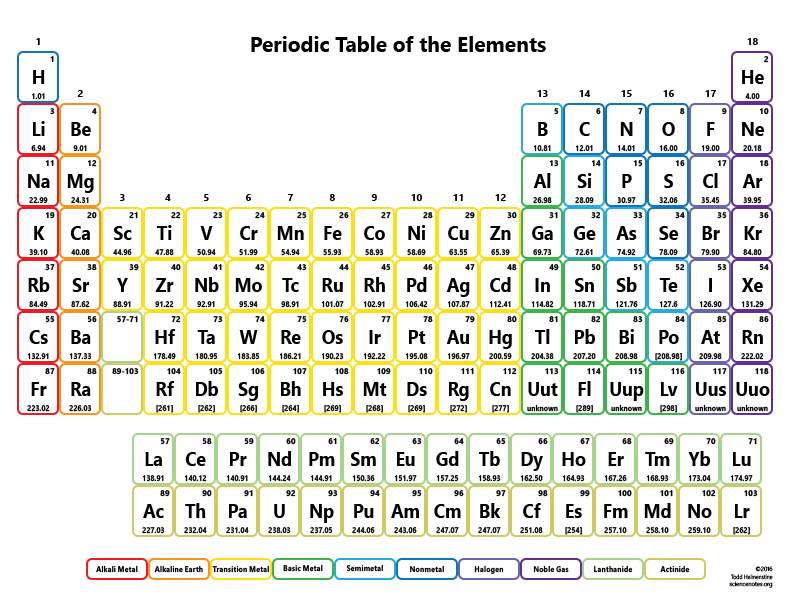
A row on the periodic table is called a period. Heres a nice Periodic Table and more pdf for you to use for this class. Element/Moleculeīy knowing the molar mass of a substance, having any mass of that substance allows one to know how many moles of it they have.\] The periodic table is your ultimate conversion chart for converting any substance into another substance and doing so with exact proper amounts (masses and moles). Masses of other atoms are expressed with respect to the atomic mass unit. When considering the element, aluminum, we can use the periodic table of elements to look up the atomic weight of this element (26.98 amu). A list of some molar masses can be seen below. The atomic mass unit (abbreviated u, altho ugh amu is a lso used) is defined as 1/12 of the mass of a 12C atom: 1 u 1 12 the mass of 12Catom (2.6.1) (2.6.1) 1 u 1 12 the mass of 12 C a t o m. In such a conversion, we use the molar mass of a substance as a conversion factor to convert mole units into mass units (or, conversely, mass units into mole units). To two decimal places, the formula mass of NaCl is 58.44 amu. The molar mass of any element or molecule is given by the sum of the atomic weights multiplied by 1 g/mol. This formula mass is the sum of the atomic masses of one sodium atom and one chlorine atom, which we find from the periodic table here, we use the masses to two decimal places: Na: 22.99 amu. The units of molar mass are grams per mole, abbreviated as g/mol. Molar mass is defined as the mass in grams of one mole of a substance. There you can find the metals, semi-conductor (s), non-metal (s), inert noble gas (ses), Halogens, Lanthanoides. The concept that allows us to bridge these two scales is molar mass. As a result of this standard, the mass of all other elements on the periodic table are determined relative to carbon-12. Please note that the elements do not show their natural relation towards each other as in the Periodic system. This is the standard unit for atomic or molecular mass, and 1 amu is thus 1/12 th the mass of a 12 C atom. The unity for atomic mass is gram per mol. In SI units, the molar mass is given by kg/ mol, however by convention the molar mass is expressed in units of g/mol. The lightest chemical element is Hydrogen and the heaviest is Hassium. The given mass of K (4. For potassium then, it is 39. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. 3: Table salt, NaCl, contains an array of sodium and chloride ions combined in a 1:1 ratio. The formula mass for this compound is computed as 58.44 amu (Figure 3.6.3 3.6. This comparison is seen below:ġ dozen = 12 objects 1 mol = 6.022141 x 10 23 objects What’s important for today’s topic is that numerically, the molar mass is equal to the average atomic mass of the given atom, so we can look it up in the periodic table. Sodium chloride is an ionic compound composed of sodium cations, Na +, and chloride anions, Cl, combined in a 1:1 ratio. When we add up the total values i.e, 46 + 12. 1 What is the molar mass of sodium carbonate, Na 2 CO 3 Solution Since sodium carbonate contains two atoms sodium, one atom of carbon and three atoms of oxygen. However when talking about moles, instead of only having a dozen, there is in fact 6.022141 x 10 23 of a given substance in a mole (known as Avogadro's number). Add up all and assign unit as grams/mole. For example, if someone has a dozen bricks and another person has a dozen feathers, they have the same amount of their substance, but a vastly different total mass. When you open any file of an element in the periodic table, you will find a small table with some basic information about that element. The periodic table lists the atomic mass of carbon as 12. Since a mole is defined as the amount of a substance and substances have different masses, each element or molecule will have a different molar mass. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. Molar mass (M) is a physical property, defined as the mass of a given element or molecule per mole of that substance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
